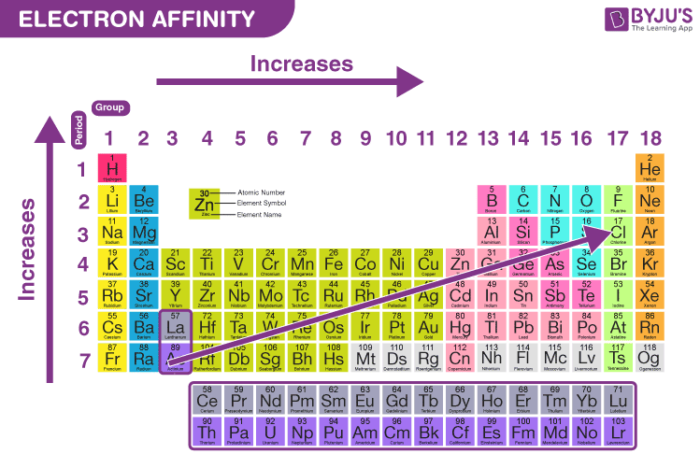
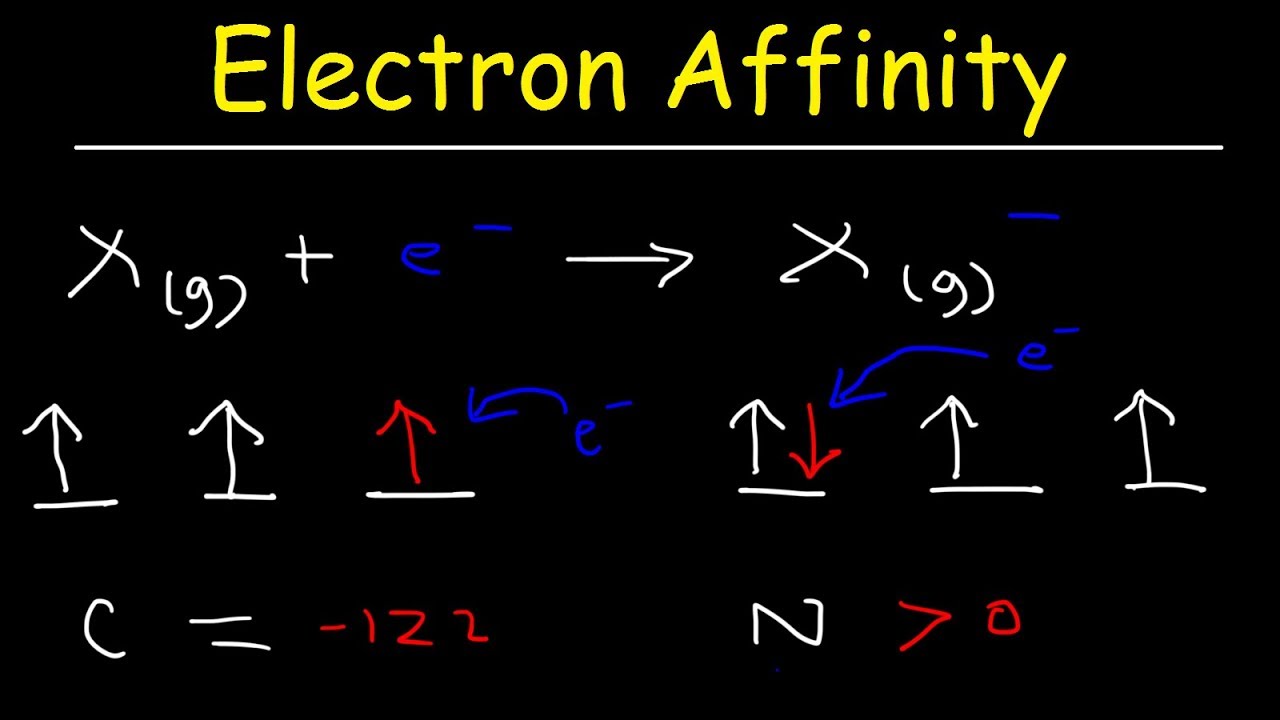
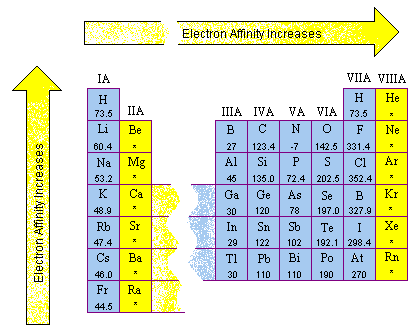
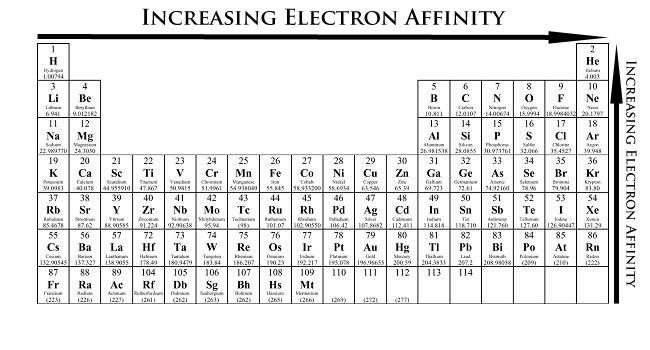
SOLVED: The first electron affinity for oxygen is +141 kJ/mol while the second electron affinity is -780 kJ/mol. Briefly account for the difference in magnitude and sign for these two values.

SciELO - Brasil - Density-functional calculations of molecular electron affinities Density-functional calculations of molecular electron affinities

SciELO - Brasil - Density-functional calculations of molecular electron affinities Density-functional calculations of molecular electron affinities

The ionization energy and electron affinity of an element are 17.42 eV and 3.42 eV respectively, find the E.N. of the element on pauling scale is:

Calculate the electron affinity of fluorine atom using the following data . Make Born - Haber's Cycle . All the values are in kJ mol^-1 at 25^∘ C . Δ Hdiss (F2) =

a) Band diagram of 2H MoS 2 . The calculated electron affinity value... | Download Scientific Diagram

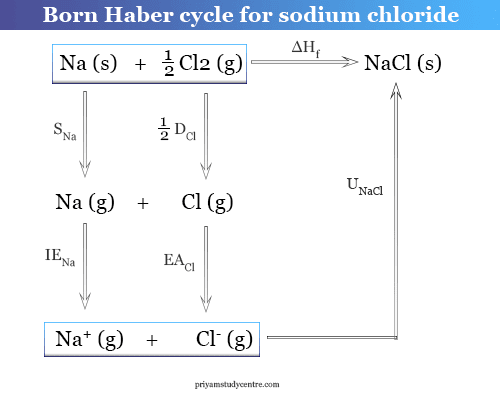
Calculate the electron affinity of chlorine from the given data: Na(g) = Na^ + (g) + e Δ H^o = 499.8 kJ 12Cl2(g) = Cl(g) Δ H^o = 120.9 kJ Na(s) =